Turn product definitions into product intelligence
QbDVision turns static definitions into real, lifecycle-driven product intelligence.
Redefine product definitions
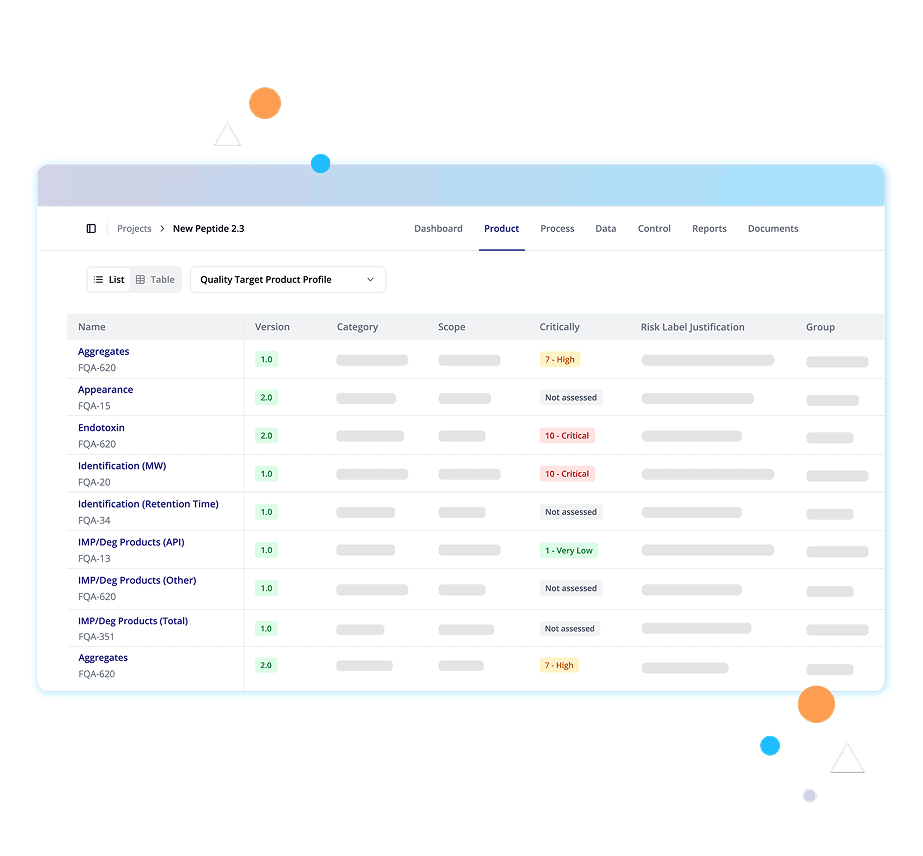
Digital Product Definition is the structured digitalization of drug product attributes, risks, and intent that turns scattered data into governed, risk-aware lifecycle control.
Structured product knowledge
Structured product data models unify Drug Substance, Drug Product, QTPP, CQAs, and Product Specifications into a governed digital definition and a reliable system of record.
Real-time risk and impact
Attribute-level risk and live traceability show how decisions impact the product before changes move forward.
Lifecycle control
Versioning and automated reporting keep product data aligned across stages with consistent, inspection-ready outputs.
Wouldn’t it be nice if your product definition was held together by more than just spreadsheets and vibes?
Product definitions are supposed to anchor quality, guide development, and defend regulatory confidence. In reality, most organizations are managing product knowledge across scattered QTPPs, CQA tables, regional specs, risk reports, and submission spreadsheets that rarely agree with each other.
QbDVision replaces document-based product definitions with a living digital system built for lifecycle control.
Why this matters
Development risk compounds
Unclear product intent and fragmented rationale create rework, misalignment, and late-stage surprises during scale up.
Hidden transfer costs
Missing context during handoffs drives delays, deviations, and repeat investigations across sites and partners.
Rising regulatory risk
Inconsistent justification and weak traceability increase inspection exposure and submission friction.
Create connected product knowledge
Digital Product Definitions in QbDVision capture the full logic behind the product, like QTPP, CQAs, specifications, risk, justification, and control strategy, in one connected environment.
Every attribute is versioned, traceable, and linked to downstream process and analytical controls, so context is never lost, even as changes are made.
The result is product knowledge that behaves like a system: inspection-ready, lifecycle-aware, and built to scale from development through commercialization and beyond.
Take control of your product definitions
Align product, process, analytical, and quality teams
Bring cross-functional teams together around a shared digital definition of product intent, risks, and control strategy, eliminating interpretation gaps and rework.

See changes across the lifecycle
Digitally trace how updates to CQAs, specifications, or acceptance criteria impact processes, methods, controls, and regulatory commitments, before changes are approved or implemented.
Manage product intent as living knowledge
Continuously refine CQAs, specifications, and rationales while maintaining complete traceability to original intent, risk assessments, and regulatory decisions.
One product definition across sites and partners
Enable collaboration across internal teams, manufacturing sites, and external partners using a single, governed digital workspace.
End-to-end product definition control
Maintain product intent, manage risk, and respond faster to regulatory questions with complete visibility across the product lifecycle.

Align teams and regions from day one
QbDVision connects global product teams, sites, and partners around a single digital product definition, eliminating ambiguity across regions and functions.
See regulatory and quality risks earlier
Understand how evolving CQAs, specs, and controls impact regulatory commitments and lifecycle strategy before submissions, transfers, or changes.
Maintain confidence as programs scale
Evolve product definitions through development, validation, commercialization, and post-approval change with full governance and traceability.
Clear product intent, predictable execution
Gain structured visibility into sponsor-defined product requirements and critical attributes, not just static documents.

Receive clearer definitions and collaborate confidently
QbDVision provides controlled access to structured product definitions, ensuring CDMOs understand not only what is required, but why.
Identify gaps early and plan proactively
Digitally align sponsor product definitions with site capabilities to surface misalignment and risk before execution begins.
Execute with confidence as changes occur
Adapt to evolving requirements while maintaining traceability back to approved product intent and justification.
The foundation of a true Digital CMC strategy
QbDVision turns scattered product documents into a structured, governed system that carries intent, risk, and control strategy across the lifecycle. QTPPs, CQAs, specs, and justifications live as connected data you can trust. Versioning is enforced. Traceability is automatic. Reporting is inspection-ready by design.
This means predictable development, faster scale-up, and less regulatory friction. Product knowledge becomes structured, connected, and lifecycle-driven. Giving everyone total visibility into the product throughout the entire process.

Share knowledge, not just PDFs
Complete, structured product definition
QbDVision captures every product detail in a structured, digitally native format. Product-specific CQAs, acceptance criteria, specifications, regional variations, DS and DP records, and attribute justifications are all connected with their scientific rationale.
Integrated risk-based criticality
Each attribute is assessed for risk, with visibility into its impact on safety, efficacy, and performance. Residual risk is tracked, and all evaluations align with ICH Q9 principles.
Regulatory & submission readiness
Structured, automated reporting and consistent product data accelerate responses to regulatory questions. Tables, reports, and submission-ready outputs are generated automatically, ensuring teams stop recreating definitions and instead manage product intent directly.
Dive deeper with our CMC thought leaders

MORE INSIGHTS
Find out how Viralgen used QbDVision to pre-build processes, enabling real-time collaboration and cutting onboarding time from 6-12 months to 3 months.

MORE INSIGHTS
The new draft guideline has been fully unveiled, and it’s truly comprehensive. Find out how it embeds Digital CMC in Modules 2 and 3.
See what Digital Product Definition looks like in practice
Talk to our Digital CMC experts today.






















































