Deliver every cure
Accelerate the journey from scientific discovery to patient delivery by unifying your CMC data into a trusted digital knowledge platform.
Trusted by 7 of the top 25 biopharma organizations and CDMOs










Who we are
We are pharma geeks who know your world, and tech innovators who see what’s possible
Today, we work with development teams to build a platform that captures structured CMC data, preserves process knowledge, and maintains continuity across the full development lifecycle.


Leading the Digital CMC movement
We’re partners who live and breathe Digital CMC. From creating community with our Digital CMC Summit to collaborating with industry leaders on Project Artemis, we’re proud to work with our partners to solve some of pharma and biotech’s biggest digital challenges.
What we do
Digital CMC for the entire product lifecycle
QbDVision’s industry-leading Digital CMC platform is built by a team of pharma experts to help biopharma organizations and CDMOs accelerate the development of life-changing therapies. Our platform provides end-to-end product lifecycle management (PLM) capabilities, from R&D to commercial manufacturing, and lays the trusted knowledge foundation for AI-driven pharmaceutical development.
Early Stage R&D
Clinical Development & Manufacturing
Commercial Manufacturing
Capacity Expansion
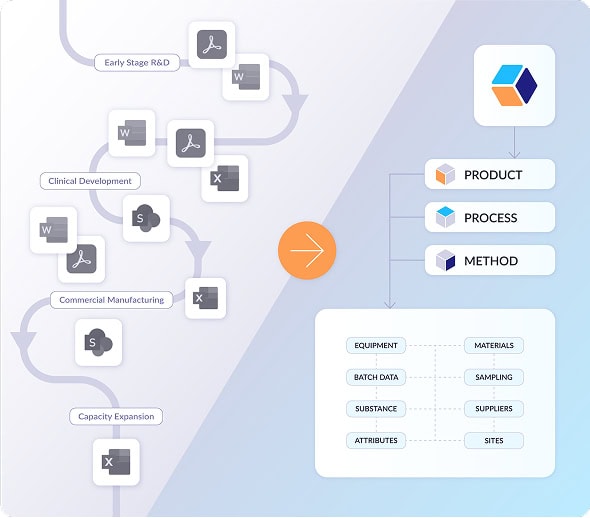
A single source of truth that unites your workflows
QbDVision enables teams to automate development workflows, streamline collaboration, and provide real-time guidance for enhanced data-driven decision-making.
Digital product definition
Build digital QTPPs, CQAs, and acceptance criteria that can be versioned, governed, and managed across the entire product lifecycle.
Digital process definition
Capture unit operations, steps, parameters, materials, equipment, in-process controls, and sampling plans.
Digital method definition
Transform your analytical methods into a living, governed, interconnected knowledge system.
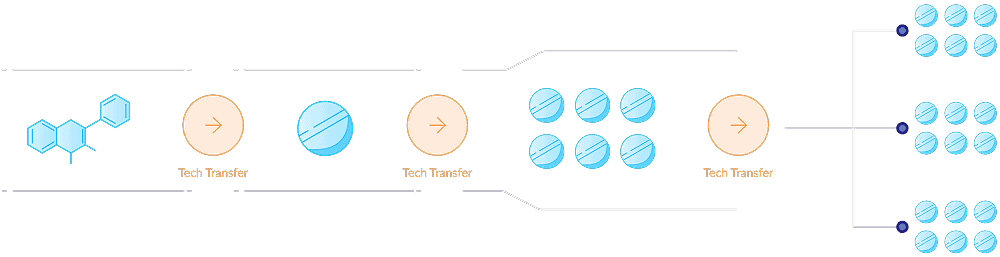
Digital tech transfer
Easily assess transfer readiness, automate key deliverables, reduce risk, and enable faster, more predictable transitions across scales, sites, and partners.
How we do it
Transforming CMC from document-first to document-free
Structured, atomized data aligned with FAIR principles is contextualized using domain-relevant taxonomies and linked via ontologies to drive consistency and efficiency. Built on a Quality by Design framework and aligned with ICH guidelines, QbDVision integrates specification and recipe management, risk management, material management, and automated report authoring—all within a validated SaaS environment.
The Digital Toolkit
End-to-end Digital CMC
100+ digital capabilities to take your team to the next level
Risk management & Quality by Design
Product & specification definition
Raw materials & supplier management
Process design & scale-up
Product & process control
Structured data & compliance
Digital Tech Transfer with facility fit
Process analytics
Automated reporting & intelligence
Knowledge & product lifecycle management
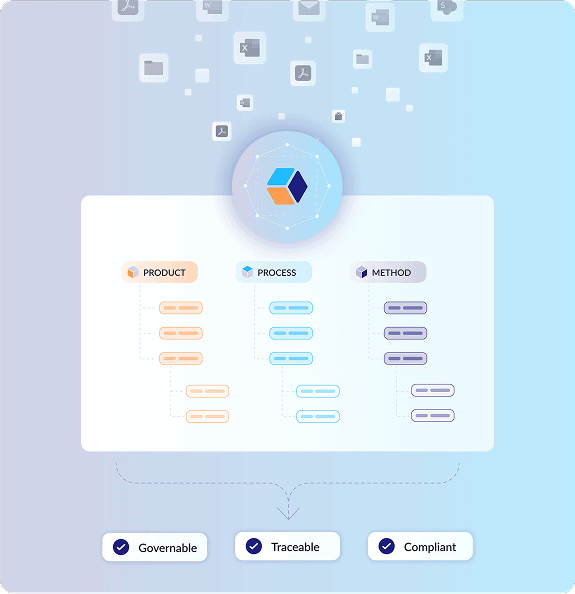
A knowledge-first foundation for AI in CMC
Our structured data framework lays the knowledge foundation for AI-powered pharmaceutical development that’s governable, traceable, and compliant.
WHY WE DO IT
To help others
Our mission is to give CMC professionals the structured digital platform they need to spend less time finding and verifying data and more time on process science and optimization. By organizing critical CMC knowledge into a single trusted source, QbDVision helps teams move therapies from development through manufacturing with greater speed and confidence. The result: faster patient access to the next generation of medicines, and a meaningful contribution to health equity.
Faster to first-in-human
Secure regulatory clearance for first clinical study faster; avoid hold for CMC deficiencies.
Faster clinical development
Reduce clinical scale to commercial scale manufacturing (tech transfer) times by 50%
Faster regulatory approval
Accelerate final approval by demonstrating robust process understanding & product quality
Faster to market(s)
Accelerate commercial launches in multiple countries
Faster capacity expansion
Increase production volume and improve supply chain resilience to meet demand

Our value
QbDVision lowers costs and accelerates time to market
80%
Reduction in tech transfer times
75%
Faster CDMO onboarding
2.5-5x
ROI for enterprise deployments
People love working with us
Check out our case studies and testimonials and see how we’re unlocking our industry’s ability to realize the full potential of its science.
95% Customer retention rate
Customer satisfaction rate
Testimonial
AskBio uses QbDVision to convert hundreds of unstructured documents into one structured model.

Case study

Viralgen uses QbDVision to cut CDMO tech transfer onboarding time by 75%.
Case study

Bayer eliminates up to 80% of tech-transfer-related meetings with QbDVision.
The secure, trusted partner for your most valuable data
Our GAMP5 Category 3 cloud-based software is compliant with ISO 27001, SOC 2, 21 CFR 11, EU Annex 11, ICH guidelines and QbD principles.






























































