The structured Digital CMC platform for end-to-end product lifecycle management
QbDVision’s category-leading Digital CMC platform is a new operating model for how CMC knowledge is structured, governed, and applied across the product lifecycle.
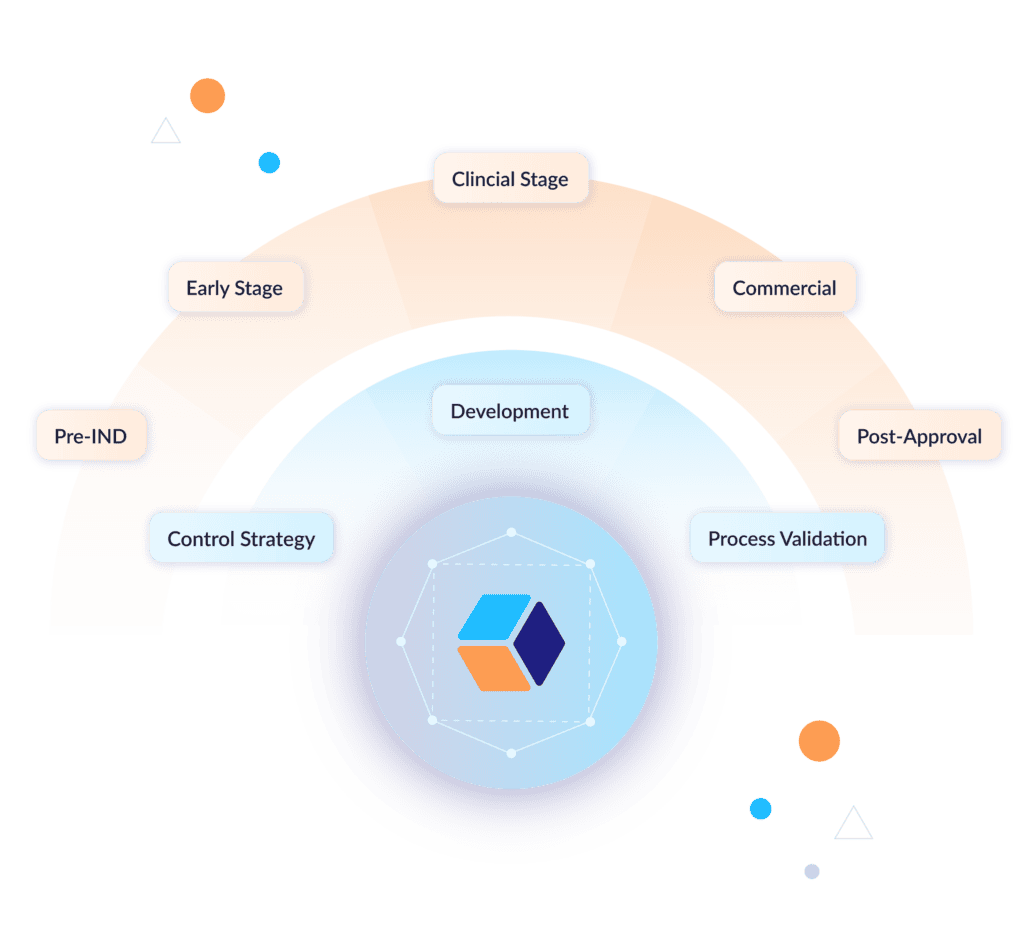
A new operating model for CMC
Chemistry, Manufacturing, and Controls (CMC) has always been a foundational function of pharmaceutical and biotech product development and manufacturing: the long-but-essential path by which safe, effective, quality medicines are delivered to patients. But the unstructured, siloed systems that have traditionally been used to manage end-to-end CMC data don’t live up to the challenges present in today’s CMC landscape.
Digital CMC provides an opportunity to move away from those inefficient systems to a more efficient digital operating model that drives transparency, collaboration, and agility throughout the entire product lifecycle.
CMC doesn’t have to be siloed
Historically, CMC data has been buried in documents and spreadsheets that are stored across file servers, quality management systems, and databases. Siloed CMC knowledge is dispersed and hard to find, data utilization and integrity are poor, preparation of regulatory submissions and tech transfers are cumbersome, and product lifecycle management is reactive.
This looks like
Disconnected folders and tools
Manual interpretation of results
Untraceable rationale and risk decisions
Hard-to-find data buried in documents
Minimal lifecycle visibility
These approaches
Lack structured context
Mask relationships between decisions and outcomes
Impede workflow integration
Prevent machine-readable governance
The first and only end-to-end Digital CMC software
Digital CMC isn’t a collection of tools, it’s a new operating model for how CMC knowledge is structured, governed, and applied across the product lifecycle. QbDVision treats CMC knowledge as connected, dimensional, and contextualized information sets that can be assembled, queried, and evolved over time, weaving previously disconnected data threads into a digital fabric.
We don’t just digitize documents or optimize isolated workflows, we digitize scientific knowledge itself. Our platform captures parameters, attributes, risks, and justifications as structured, connected, lifecycle-aware knowledge, creating a single, governed source of truth that spans product, process, method, transfer, validation, and post-approval change.
Built from the ground up to align with ICH Q8–Q12, Q14, and ALCOA+ principles, QbDVision enables organizations to move from document-driven execution to a scalable, inspection-ready, and future-proof Digital CMC operating model.

The keys to Digital CMC
Knowledge-first architecture
Organize technical data, parameters, attributes, and relationships into connectable entities. This converts isolated outputs into a knowledge fabric that teams can trust and reuse.
Lifecycle integration
Ensure that information flows seamlessly from early discovery through development, tech transfer, validation, and commercial operations while maintaining context and traceability.
Digital process orchestration
Capture workflows in digital formats that can be tracked, automated, and analyzed, including risk assessments, justifications, and decision logs.
Information governance and standards
Define taxonomy, metadata standards, and governance protocols that ensure data integrity and regulatory readiness.
Ecosystem integration
Connect Digital CMC capabilities with systems like LIMS, ELN, MES, and QMS, to extend visibility and deliver actionable insights.
Make your document-based workflows history
Our approach to CMC puts people at the center of the process, helping you move away from inefficient, document-centric frameworks and toward efficient digital structures that drive collaboration and transparency and minimize rework.
Before QbDVision
Siloed documents
Manual, untraceable decisions
Poor knowledge reuse
Slow coordination and rework
With QbDVision
Connected knowledge architecture
Contextualized, traceable decisions
Lifecycle visibility and reuse
Scalable workflows and reduced risk
Digital CMC unlocks enterprise value
Faster to first-in-human
Faster clinical development
Faster regulatory approval
Faster to market(s)
Faster capacity expansion

This is not a moment, it’s the movement
Digital CMC transforms CMC from a set of legacy workflows into an integrated, knowledge-driven, lifecycle-first engine for high-performance drug development.
It unites structured knowledge, lifecycle context, collaborative processes, and connected systems and modernizes how technical development functions, how teams work together, and how decisions are made across modalities and stages.
Digital CMC is the future, and the future is already here.
“With Digital CMC, we’re not only reimagining how program data is captured, organized, and curated, we’re reimagining how people collaborate within organizations and how we in the broader CMC community collaborate with each other.”
Dive deeper with our CMC thought leaders
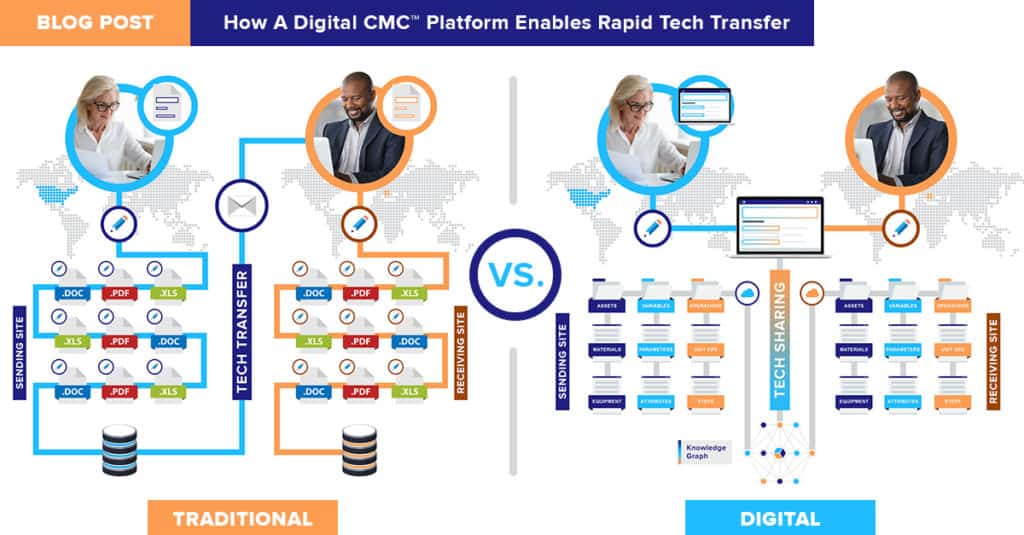
MORE INSIGHTS
Take a deep-dive with QbDVision Co-Founder & CEO, Yash Sabharwal.

MORE INSIGHTS
See how Bayer digitized a complex tech transfer with QbDVision, accelerating timelines, improving collaboration, and ensuring regulatory confidence.























































