- Flagship platform for pharmaceutical process development and manufacturing expanded to streamline compliant workflows with integrated functionality
- First-of-its-kind structured knowledge management solution supports industry transformation aligned with Pharma 4.0 initiatives and KASA (FDA)
- Risk-based emphasis accelerates development with smarter resource allocation and improved manufacturing efficiency
- Robust data importation, knowledge graph and risk management tools verified through multiple independent security and compliance parties, and validated for GMP use
AUSTIN, Texas — June 18, 2019 — CherryCircle Software, the company providing the QbDVision® platform to accelerate the development of manufacturing processes for the pharmaceutical industry, announced today significant new features, including process capability analytics, expanded support for Contract Development and Manufacturing Organizations (CDMOs and CMOs) and consultants, and data import capabilities for faster project setup and data ingestion. QbDVision is the first-of-its-kind, cloud solution for risk-based management of pharmaceutical process development. The platform captures and aggregates drug development knowledge over time for smarter development within a single, validated solution.
“We are fundamentally changing how the pharma and biotech sectors approach product life cycle management,” said Yash Sabharwal, President and CEO, CherryCircle Software. “The antiquated solutions used today inhibit holistic process development that would speed drugs to market with lower manufacturing costs. Development is becoming increasingly more complex with new, small-batch cell, gene and curative therapies, and this will require smarter and more efficient manufacturing. QbDVision is enabling knowledge and quality risk management over the development lifecycle, revamping how the drugs of tomorrow are advanced today.”
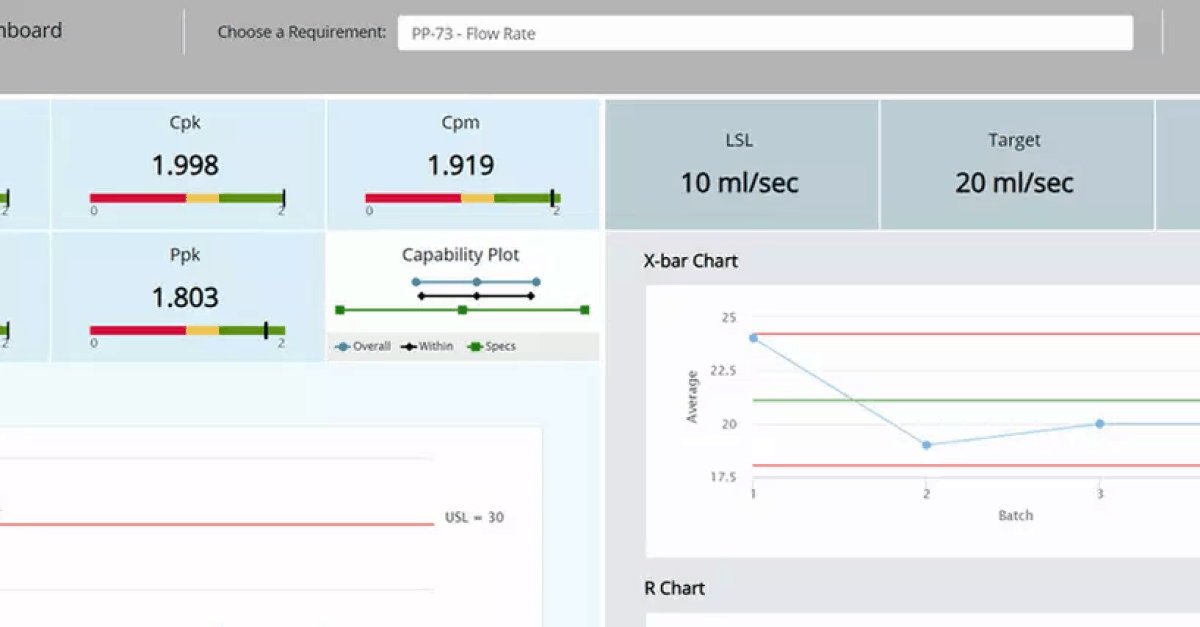
With over 100 new capabilities and improvements created through active customer, partner and adviser feedback, QbDVision unites robust data importation tools with novel visualization, risk management and analytics for assessment of process capability within a 21 CFR 11 environment for the most powerful structured knowledge management application available.
“These recent platform enhancements advance our mission to help pharma companies deliver the highest quality pharmaceutical therapies to market faster and at a lower cost with better design and control of complex manufacturing processes,” said Sabharwal.
Features of the updated platform include:
- New import functionality – Existing projects and processes set up faster by directly importing project records, suppliers and users with Excel. Track and manage process performance data by uploading batch data or continuous data from manufacturing runs.
- Integrated data analytics – Automate process capability analytics of manufacturing data. New functionality allows end users to automatically calculate KPIs and support data-driven decisions about process definition and control.
- Improved reporting – New reporting capabilities allow customers to visualize and work with data across systems.
- Support for contract manufacturers (CDMO/CMO) and consultants – Amplified collaboration capabilities make it easier to work in teams, and project-level security permissions provide specific access control. Features expanded for creating templates and cloning existing records for platform-level process management.
The Mandate to Modernize Pharmaceutical Drug Development and Manufacturing
Biotech and pharma manufacturers have historically used multiple, poorly-integrated software tools for knowledge management. This approach has hampered risk-based process development and compromised data integrity. This is one of the many facets contributing to an average of $2.5 billion and 11 years for a drug to be created and sold
QbDVision provides a streamlined knowledge management structure that can improve the quality of drugs by unifying and simplifying the product management process. It provides an integrated repository to preserve knowledge; track the evolution of risk based on data and scientific rationale; improve manufacturing efficiency; and help constituents navigate the changing regulatory environment.
Tailored for process engineers, chemists, technical operations teams and quality/regulatory groups in the biotech and pharma sectors, the platform employs serverless technologies and automated testing tools for the rapid development and deployment of secure, validated solutions that enable process and data management under a quality-by-design (QbD) framework with minimal IT burden.
QbDVision is available for a free 30-day trial. For more information, reach out about exploring these new capabilities.
About CherryCircle Software, Inc.
CherryCircle Software, Inc. builds cloud-based solutions to help the pharmaceutical industry reduce the time and cost of development, and increase the availability and affordability of essential therapies for patients. Its flagship platform, QbDVision®, provides the pharmaceutical industry with simple, yet powerful tools to facilitate robust manufacturing process development and product life cycle management driven by data and science, enabled by people, and powered by innovation and automation. Founded in 2016 by pharmaceutical manufacturing and quality experts, CherryCircle is headquartered in Austin, Texas, and is an active member of the International Society for Pharmaceutical Engineering (ISPE). For more information, visit our website and follow us on Twitter.
Media contact
Gina Manassero
(512) 553-2387
gtmanassero@gmail.com